Catching osteoarthritis early
Cartilage covers the surface of joints preventing damage to soft tissues whilst supporting the body’s weight during movement and exercise. However, the structure and function of cartilage is often damaged by injuries to the knee joint and the risks increases with age due to changes in tissue function. In the long-term, the normal functionality of cartilage changes as a person gets older and protein malfunction could make the tissue more prone to damage and lead to cartilage disorders such as osteoarthritis.
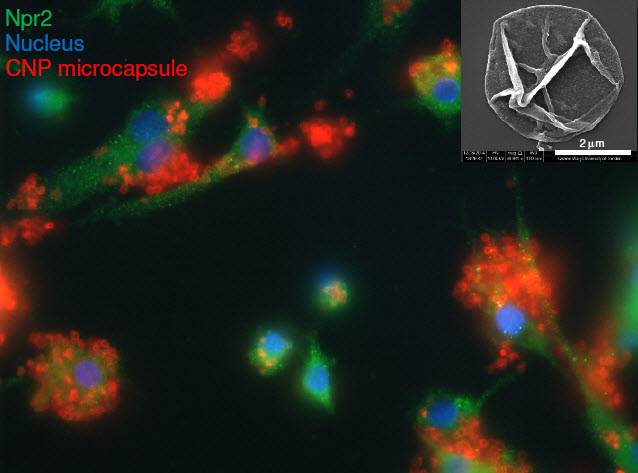
We have evidence that a molecule called C-type natriuretic peptide (CNP) occurs naturally in human cartilage and activates a novel mechanism that maintains normal protective function and encourages the growth and repair of cartilage. CNP promotes cartilage growth and prevents the onset of damage, by activating two proteins called Npr2 and Npr3 which are found on the surface of cartilage cells called chondrocytes. Although CNP can bind to either of these proteins, previous studies reported that their activation has different effects on the cells behaviour. We identified that CNP promotes beneficial protective effects on cartilage growth when it interacts with Npr2. In contrast, Npr3 was thought to reduce signalling via Npr2 therefore dampening the protective effects induced by CNP. However, our published studies provide evidence that treatment of human cartilage with agents that specifically activate Npr3 was found to enhance the beneficial effects of CNP such that the protective and reparative response was augmented with mechanical loading.
Our findings have been highlighted by Arthritis Research UK as an exciting discovery and imply that Nprs are key molecules in maintaining normal cartilage function and mediate the beneficial response to mechanical loading.
However, the local delivery of therapeutics for the treatment of osteoarthritis has been challenging. Current treatment options are suboptimal for most patients due to poor targeting of therapeutics to the pathological site to maximise efficacy and reduce side effects. Drug costs are also high.
Emerging strategies including drug-delivery microcapsules for cartilage offers promise since the therapy improves delivery to the tissue and will expose cells to higher drug concentrations.
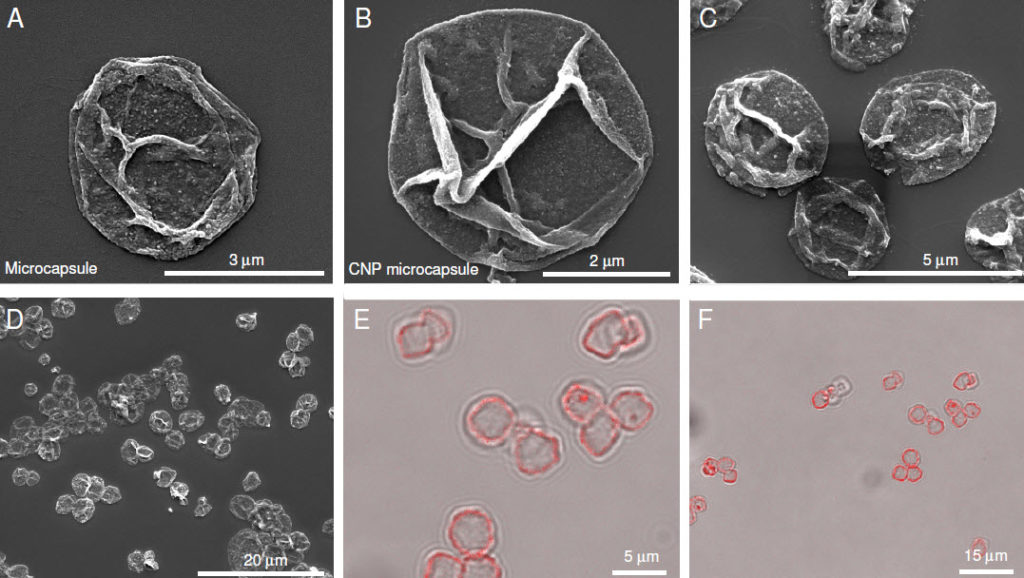
For example, our recent data published in the journal Biomacromolecules (http://pubs.acs.org/doi/abs/10.1021/bm501575w), provides evidence that CNP microcapsule technology is effective in preventing matrix loss and reduced the inflammatory disease process in an animal model.
The CNP microcapsule technology has the potential to be highly attractive to investors because it represents patentable technology to augment a natural pathway with known beneficial effects in cartilage, bone and cardiovascular tissue. Our work will help others develop treatments such as preventative CNP-based medications and in the long term, improve clinical therapies. Selective agonism of CNP signalling could be an effective age-linked target to promote health and wellbeing in older people.